Life Science Factory @ Festival der Zukunft
Improving the world – but how? We partnered up with the Festival der Zukunft for the third time to find out how we can shape our future with technology, science and creativity.
We’re excited to dive into transformative topics in life sciences and entrepreneurship. Join us if you want to explore:
Friday // 13:45 // Theater Stage
Panel: “Can Artificial Intelligence Finally Cure Cancer?” – featuring our community members Christina Port, 2NA Fish and Daniel Soric, Deep LS
Friday // 14:30 // orange room
Life Science Meet-up: Connect and learn within a creative networking space where innovation thrives and ideas come to life – hosted by our Venture Team Martin Strehle, Irina Reimer and Max Zinowsky
Friday // 16:00 // orange room
about the life science factory
vision.
facilities.
programs.
benefits.
Benefits for investors

- Mitigate Risks and Enhance Outcomes: By integrating your portfolio start-ups into the Life Science Factory, you leverage our first-class infrastructure and expertise, significantly enhancing the probability of their success.
- Access to Expert Guidance: Start-ups benefit from continuous support from our specialists, particularly through the critical stages of fundraising and business development, which can improve their stability and growth potential.
- Quality Assurance in Investments: Through our meticulous screening and selection process, we ensure that only the most promising and qualified start-ups are presented to investors, thereby minimizing investment risks.
- Early Investment Opportunities: We provide exclusive early-stage investment opportunities, allowing you to invest in promising start-ups before they catch the broader market’s attention.
- Reliability and Credibility: As an affiliate of Sartorius AG, we stand as a trustworthy partner, ensuring a stable and reliable collaboration for both investors and start-ups.
Benefits for start-ups
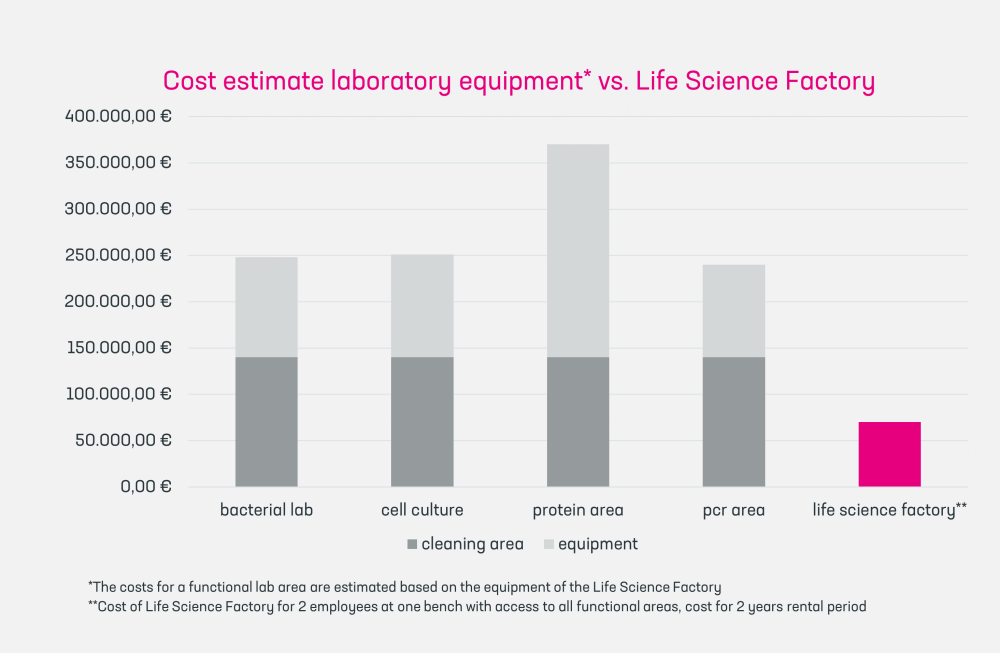
- Advanced R&D Capability: We provide state-of-the-art lab equipment with supervision, allowing start-ups to focus on innovation without the overhead of equipment management.
- Branding and Networking: Through collaboration, we enhance the reputation and visibility of start-ups, expanding their network and opening up new opportunities.
- Operational Flexibility: Our shared lab benches and flexible coworking spaces offer a quick start and scalable options to accommodate the changing needs of start-ups.
- Regulatory and Pre-operational Support: We support start-ups with essential services before the rental period begins, including communication with authorities, easing the regulatory setup process.
- Knowledge Exchange and Skill Development: Our interdisciplinary teams and alumni network deepen know-how and facilitate exchange, enriching the life science community’s collective expertise.
- Long-term Growth and Connections: We connect founders with relevant experts and partners, and provide referrals to accelerator programs, ensuring sustained growth and benefits from our network.
- Educational Opportunities and Community Engagement: Start-ups benefit from free participation in our programs and events, promoting continuous learning and engagement within the community.
discover the life science factory
Explore our facilities in Göttingen with our virtual tour. Step into our innovative spaces designed for collaboration and progress, and explore all we have to offer.
Discover our new facilities developed in collaboration with Helmholtz Munich in Munich Neuherberg. This innovative hub provides start-ups with laboratory spaces and access to extensive support services.
Lets Connect
Contact Us
Meet us @HTGF Family Days
May 11, 2026 — May 12, 2026 / 8:00 am
The HTGF Family Day is the most important portfolio event of High-Tech Gründerfonds (HTGF) – a two-day industry gathering that brings together up to 1,500 people from HTGF portfolio startups, investors, companies, and ecosystem partners in Berlin to shape the next chapter of Europe.
Meet us @Lift-Off
May 28, 2026 / 6:00 pm
The LIFT-OFF start-up competition at the University of Göttingen is aimed at both those interested in starting a business and active founders from all disciplines. It is accompanied by workshops, events, and a mentoring program in the field of entrepreneurship. In addition to invaluable inspiration and experience, there are also attractive cash and non-cash prizes to be won, which are awarded by a jury. We are delighted to support the competition with our sponsored special prize, “Life Science.” The award ceremony will take place on May 28, 2026.
meet us @bio:cap
June 09, 2026 — June 11, 2026 / 1:00 pm
Premiere of a new Life Science & AI Investival at CityCube Berlin. With bio:cap, a new international event format launches in 2026, bringing Life Sciences and Artificial Intelligence together on one platform. The first edition will take place 9–11 June 2026 at CityCube Berlin. bio:cap combines an expo, conference, and curated networking, bringing together stakeholders from biotechnology, pharma, diagnostics, TechBio, data & AI, as well as investors, research, clusters, policymakers, and innovation ecosystems.
meet us @Investordays
June 11, 2026 / 2:00 pm — 10:00 pm
Investor Days Thuringia (IDT) on 11 June 2026 is a leading pitch and networking event in Erfurt that connects early-stage startups with investors, corporates, and research institutions.
Selected startups present their business models, while targeted 1:1 meetings and networking sessions foster investment opportunities and strategic partnerships.
ELSA Pitch Bowl
June 25, 2026 / 5:30 pm
The final event of the Entrepreneural Life Science Accelerator program.
In 3-minute pitches, the founding teams present their development in the areas of idea, market, and team, and receive on-stage feedback and critical questions from an experienced investor jury, which awards a prize to the most promising team. A joint closing with snacks and drinks rounds off the evening and will offer space for exchange and networking.
Mentor Match Meetup
September 17, 2026 / 4:30 pm
Our Mentor Match program is continuing successfully and is supported by our partners IHK Göttingen, the University's Transfer & Startup Hub, and the Life Science Factory. At our annual Mentor Match Meetup on site, it's time once again to deepen the dialogue and look back on successes achieved through innovation, expertise, know-how, and above all, entrepreneurial spirit.
The focus is on networking and personal exchange: We are pleased to invite start-ups and corporate partners to our Mentor Match Meetup, which will take place on September 17, 2026, starting at 4:30 p.m. at the Historic Observatory Göttingen (Geismar Landstraße 11).
The agenda includes a unique opportunity to get to know the members of our 1:1 matching concept even better and to make valuable contacts. We also offer exciting ideas and workshops on the topic of entrepreneurship.
Meet us @BioEurope
November 09, 2026 — November 11, 2026 / 8:00 am
BIO-Europe, Europe's leading partnering event for the life sciences industry, will take place in Cologne from November 9 to 11, 2026. Around a thousand experts from the fields of biotechnology and pharmaceuticals, as well as investors from all over the world, will come together to form innovative partnerships and discuss industry trends. The event offers inspiring keynotes and panel discussions, as well as numerous opportunities for one-on-one meetings via the proven partneringONE system.
By displaying this Google map you agree to Google’s use of cookies. This may include analytics, personalization and ads.
Learn more



